Respiratory syncytial virus (RSV)’s high burden has made the research and development of RSV vaccines a global health priority.
RSV is the leading cause of viral lower respiratory tract infection (LRTI, including bronchiolitis and pneumonia) worldwide. Every year, an estimated 33 million children under age five are infected with RSV-associated LRTI, leading to 3.6 million hospital admissions and to 26,000 in-hospital deaths. Nearly all (95%) of these infections occur in low- and middle-income countries (LMIC). RSV also affects 1.5 million adults aged 65+ years every year, resulting in an estimated 336,000 hospitalizations and 14,000 deaths.
Looking at the economic impact of RSV, in the UK, healthcare costs and productivity losses resulting from RSV in young children are estimated to be £80 million annually. In South Africa, the estimated mean annual cost of RSV-associated illness in children under age five was US$137 million (2011-2016). A study focused on 71 Gavi countries found that the RSV-associated disease burden among children in these countries is estimated at an average of US$611 million discounted direct costs.
Treatment for RSV is limited to therapeutic measures to prevent dehydration, improve oxygen flow, and reduce symptoms such as fevers. However, in the past decade, there have been significant advances in the research and development for direct treatment as well as prevention through monoclonal antibodies and vaccines. Earlier this year (2023), the FDA and EMA approved the first-ever RSV vaccines for adults, and in July, the FDA and EMA approved the first vaccine for pregnant women to protect their babies against RSV during their first six months after birth.
Given the urgent and unmet public health need of RSV, as well as the new availability and forthcoming launch of several vaccines, there is renewed focus on ensuring we understand the multidimensional aspects that influence vaccine uptake. Overcoming confidence, complacency and convenience barriers – key determinants of vaccine hesitancy – is critical to ensure protection, especially for those most vulnerable.
“RSV is a major reason why babies end up in hospital, and it can be a lethal virus anywhere in the world. To have an effective vaccine that can be given to the expecting mothers before the babies are born is a great way of preventing this complication.” – Beate Kampmann, Professor of Paediatric Infection & Immunity and Director of the Vaccine Centre at the London School of Hygiene and Tropical Medicine
“We’re sort of going from famine to feast. These are huge game changers for what has typically been the most common cause of hospitalization for young infants in the U.S.” – Dr. Kristin Moffitt, infectious-disease specialist at Boston Children’s Hospital
Decisions on vaccine uptake are complex.
Immunization is well recognized as one of the greatest public health interventions of all time. However, the value of vaccines can only be realized if the target populations have easy access to the vaccines and are willing to be vaccinated.
Recent data shows that vaccine hesitancy was accelerated by the COVID-19 pandemic and that it will have a system-wide impact on uptake of routine immunizations. The perception of importance of vaccines is declining, their safety and efficacy are being questioned, and an individual’s social and environmental context is playing an increasing role in driving intent to get vaccinated.
In particular, the decline in positive vaccine perception in people under 35 and among women is concerning given their important role in advancing vaccine uptake in newborns, children, and their elderly parents.
An opportunity exists to shape the environment now and lay strong foundations for successful RSV vaccine introduction.
The global RSV vaccine development pipeline and the growing concerns of increasing vaccine hesitancy gives urgency to proactively shape the environment to prepare for the successful launch, roll-out and uptake of the long-awaited RSV vaccines.
The decision to get vaccinated is complex and multi-dimensional due, in part, to the interaction of different social, cultural, and personal factors. Some of the determinants of individual decision-making regarding vaccination include disease and vaccine awareness, disease risk perception, past experiences with other vaccines and/or engagement with the health system, healthcare provider advice, and ease of accessing the vaccine, among others. Many research frameworks exist to identify and map the drivers and considerations that guide these decisions.
The 3C (confidence, complacency, convenience) framework provides a lens to identify, map and analyze the factors that influence vaccine uptake in order to support the development of tailored interventions to address key barriers across target populations.
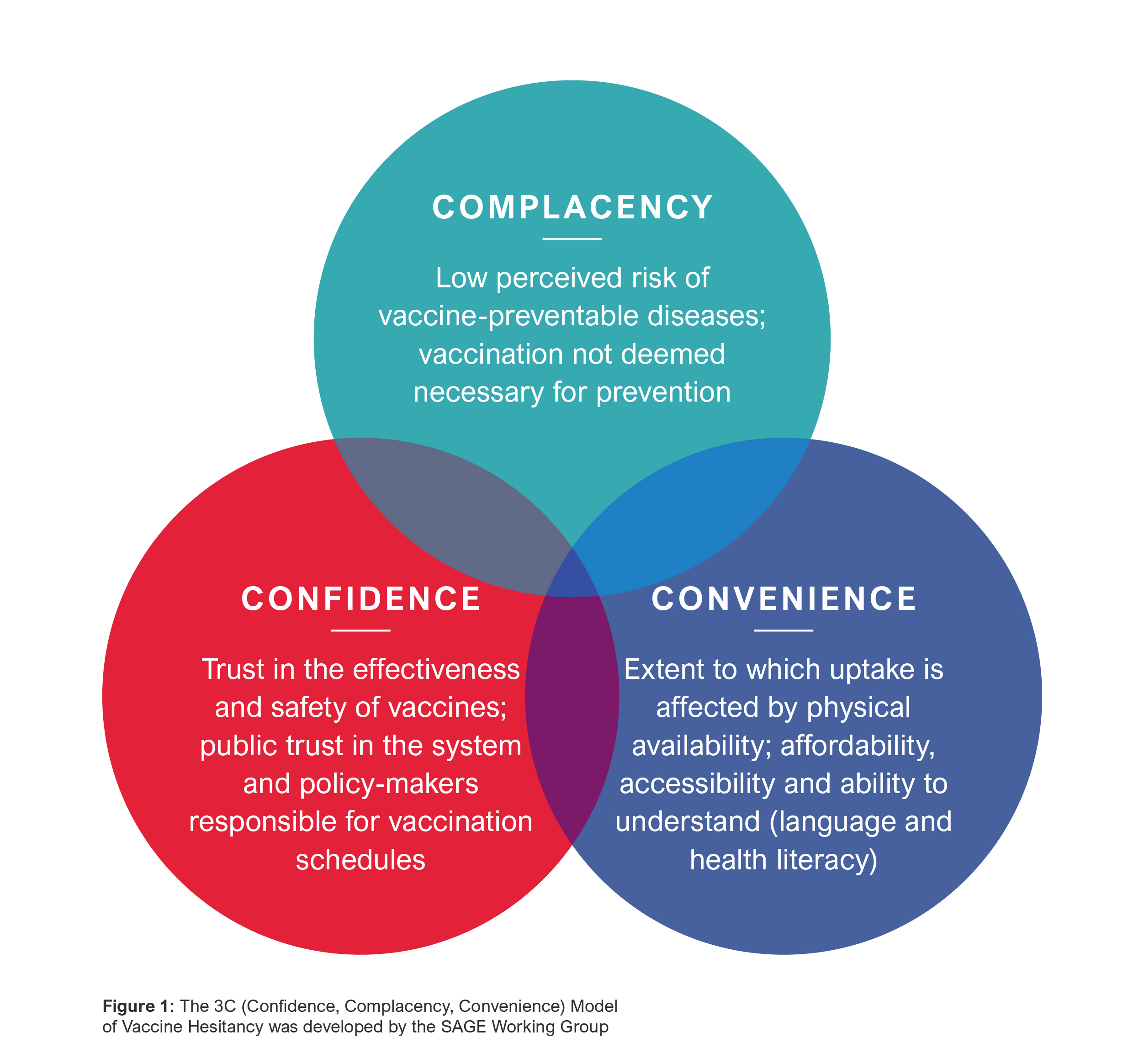
An understanding of these individual drivers and considerations, which are hyper-localized, is key to developing policy, public health and commercial strategies that will help shape RSV vaccine introduction efforts and planning for positive public health impact.
Based on our deep expertise in the vaccines space as well as our work to strengthen health systems, including through community-based approaches, Rabin Martin believes that the greatest barriers limiting the uptake of RSV vaccines will be complacency – the threat of RSV is still inadequately understood by parents and caregivers – and convenience in access to vaccines due to structural challenges of health systems and existing social-cultural barriers, especially for 60+ (adult immunization infrastructure is less developed than that for children), and infants born when RSV is not in its peak season (who will require additional touchpoints with the health system).
As such, targeted approaches that mitigate these barriers and reach the populations for whom the vaccines are intended is crucial. Continued confidence-building and health system strengthening to underpin the below strategies is also important in maximizing reach and public health impact.
| Target Population | Drivers and Considerations | RM Insight |
| Women of childbearing age (WoCBA) for maternal vaccines |
|
|
| Parents & Caregivers for pediatric vaccines |
|
|
| Adults 60+ years for older adult vaccines |
|
|
All stakeholders must anticipate vaccine introduction to drive uptake and positive public health outcomes.
Looking forward, the global health community, including manufacturers, governments, multilateral organizations and non-governmental organizations, have an ongoing responsibility to anticipate vaccine introduction challenges and develop holistic strategies to improve public health before and after vaccine launch. This is especially the case when new mechanisms of action are introduced and when vaccines will be provided outside of the routine schedule.
At Rabin Martin, we support our clients to address the threat of vaccine hesitancy, build trust, and prepare for vaccine introduction to bring positive health impact.
Together we can:
- Advance global, regional and national policies especially in LMICs to facilitate access to vaccines and ensure implementation within existing immunization systems and structures while accounting for the complexity of RSV implementation (before/during the RSV season)
- Develop localized, tailored strategies, programs and partnerships to address the practical barriers to vaccination that are most relevant to target populations (incl, WoCBA, parents and caregivers, and older populations) including by integrating community-based approach and addressing social determinants of health.
- Integrate communities’ perspectives and lived experiences to propose sustainable inclusive approaches and tactics that improve messaging on vaccination, advance confidence in the benefits of vaccines, and contribute to the design of service delivery strategies.
- Design interventions to empower health providers with adequate knowledge of RSV burden and vaccines, so that they can address caregiver and vaccine recipient concerns and confidently recommend vaccination.
- Leverage innovative approaches through meaningful partnerships with local, regional and global organizations (e.g., behavior change interventions, including through social media) to address the three C’s.